- Autor:
- Eduardo Anitua
- Mohammad H. Alkhraisat
- Asier Eguia
- Allende M
Bone-Regenerative Ability of Platelet-Rich Plasma Following Sinus Augmentation with Anorganic Bovine Bone: A Systematic Review with Meta-Analysis
Abstract
Background: The objective of this systematic review is to assess the effect of the adjuvant use of platelet-rich plasma (PRP) and its type on new bone formation by anorganic bovine bone during maxillary sinus floor augmentation procedure.
Methods: PubMed, Cochrane Central Register of Controlled Trials, and Ovid databases were searched for relevant studies published up to 16 September 2021. Randomized clinical trials (RCTs) and non-randomized controlled clinical trials (CCTs) that reported data on the new bone formation (measured by histomorphometric analysis) were considered. Risk of bias and quality assessment of included studies were evaluated following the Cochrane Handbook for Systematic Reviews of Interventions and the Risk Of Bias In Non-randomised Studies of Interventions (ROBINS-I) tool. Strength of evidence was assessed following the approach of the Agency for Healthcare Research and Quality (AHRQ) through its evidence-based practice center (AHRQ EPC). The meta-analysis was based on the primary outcome of newly formed bone, for which the standard mean difference was calculated.
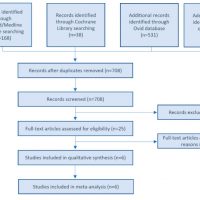
Results: After the application of eligibility criteria, six clinical trials (three RCTs and three CCTs) covering 85 maxillary sinus floor elevation procedures were included. The pooled new bone formation value for PRP was 1.67 (95% CI: -0.15 to 3.49; I2: 86%), indicating the absence of significant effect. Plasma rich in growth factors (PRGF) was the pure PRP tested in five of the included studies. When sub-group (type of PRP) meta-analysis was performed, significantly higher new bone formation was observed in the PRGF group [2.85 (95% CI: 0.07 to 5.64; I2: 88%)] in comparison to the control group.
Conclusions: A beneficial effect on new bone formation after maxillary sinus floor elevation can be obtained when anorganic bovine bone is mixed with PRGF.

 English
English
 Français
Français
 Deutsch
Deutsch
 Italiano
Italiano
 Português
Português

![Figure 2 Quality assessment of the included RCTs. (a) Risk of bias summary, review of authors’ judgments about each risk-of-bias item for each included study. (+): low risk of bias. (?): unclear risk of bias. (b) Risk-of-bias graph, review of authors’ judgments about each risk-of-bias item, presented as percentages across all included studies [36,38,40].](https://www.btitrainingcenter.com/wp-content/uploads/2022/10/bioengineering-09-00597-g002-200x200.jpeg)
![Figure 3 Quality assessment of the included prospective non-randomized studies. (a) Risk-of-bias summary, review of authors’ judgments about each risk-of-bias item for each included study. (+): moderate risk of bias. (b) Risk of bias graph, review of authors’ judgments about each risk-of-bias item, presented as percentages across all included studies [8,35,47].](https://www.btitrainingcenter.com/wp-content/uploads/2022/10/bioengineering-09-00597-g003-200x200.jpeg)
![Figure 4 Meta-analysis of the studies evaluating new bone formation. (A) Randomized clinical trials and controlled clinical trials. (B) Only randomized clinical trials. SE: standard error. SMD: standardized mean difference. CI: confidence interval [35,36,38,40,47].](https://www.btitrainingcenter.com/wp-content/uploads/2022/10/bioengineering-09-00597-g004-200x200.jpeg)